 
The difference between an emulsifier and a surfactant. These hydrophobic groups are typically at the ends of the molecule and form what's called a hydrophobic tail. These different actions are achieved by different functional groups in the molecule! In short hydrophilic functional groups are attracted to water while hydrophobic groups are attracted to oil. This means that they have hydrophilic and hydrophobic parts of their molecular structure. To have this ability emulsifiers have to be able to interact with water and oil, as I stated above. The opposite is true for Water-in-Oil Emulsions the emulsifier surrounds the water in these solutions. 
So for Oil-in-Water Emulsions the emulsifier creates a sphere around the oil. They do so by creating a sphere around whatever phase represents the smallest fraction in the final solution. This allows them to interact with both phases to create a semi-stable solution. An emulsifier is a compound that is attached to both oil and water. Now that you know what an emulsion is we can get into how an emulsifier is used to make them. Below are several more examples that you see on a daily basis. Examples of natural emulsionsĪbove I gave you some examples of different emulsions but really the list is endless. 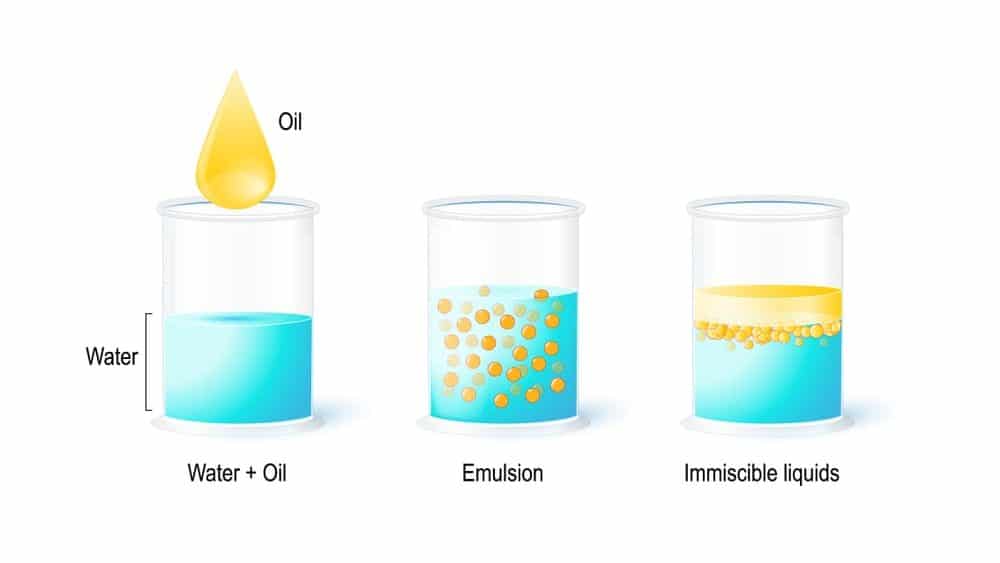
What makes these even more versatile is changing the type of oil, or even butters, used. You see, when you change the amount of water or oil the final consistency of your emulsion completely changes. Pretty easy to remember right?! An example of this type of emulsion is butter! In butter the oil phase consists of fat solids that give butter its solid form but the small addition of water makes it creamy! If more oil is used it is called a Water-in-Oil Emulsion (W/O). A great example is milk! This type of solution is usually very thin and has the physical characteristics of water. In these a small amount of oil is dispersed in water. When more water is used the emulsion is known as an Oil-in-Water Emulsion (O/W). There are two main types of emulsions depending on if there is more water or more oil in the final solution. This is impressive when you think about how water and oil do not usually dissolve in one another! This means the oil and water are combined to create a stable solution. Emulsifiers allow for the creation of emulsions! Makes sense right? Basically, these are a combination of oil and water that does not separate. Before we can talk about what an emulsifier is, first let's talk about what they create. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
